A 28 year old gentleman was involved in road traffic accident. He had been trapped under a car for several hours prior to being extracted by the fire service.
He sustained bilateral injuries to his legs with femoral and tibial and fibular fractures. Following his injuries he was catheterised and an accurate fluid balance was undertaken.
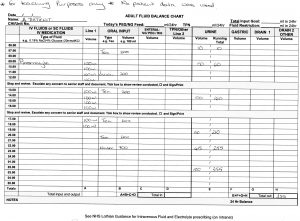
3 Days following admission following re-introduction of food and fluids the fluid balance above was taken.
Total fluid in in 24 hours 2500ml, output 355ml
Balance = + 2145ml
As above it was noted that his urinary output was low, despite an apparently adequate fluid intake.
|
Hb 114 WCC 10.2 Plt 234 K 6.4 Na 136 Urea 22.0 Creat 320 eGFR 21 |
[H+] = 50 nmol/L pH = 7.3 PaCO2 = 3.2 kPa [HCO3] = 12 mmol/L |
What does this ABG reveal about this patient’s blood picture?
(choose one of these and then click to see if you are correct)
This is metabolic acidaemia with partial respiratory compensation. The metabolic acidaemia is due to renal failure.
Typical metabolic alkalaemia presents with increased HC03 – followed by a small compensatory PC02 increase.
Typical respiratory acidaemia presents with increased PaC02 and compensatory increased [HC03-].
The PaC02 is low, which occurs in respiratory acidaemia, but in this case the low PaC02 is due to compensatory response to metabolic acidaemia.
Consider the clinical circumstances. [HC03-] is lowered which indicates acidaemia but the drop in PaC02 is a compensatory effect due to hyperventilation.
What is the cause of his acid base disturbance?
(choose one of these and then click to see if you are correct)
Myoglobin is less soluble in more acidic conditions and precipitates in renal tubules leading to renal impairment and possible kidney failure.
In rhabdomyolysis muscle cells are damaged with the release of their content. Symptoms related to this may include muscle pain and reddish/brown discolouration of urine.
This would cause respiratory acidaemia.
This is unlikely and you would tend to see peripheral signs of overload (peripheral oedema etc) prior to this occurring.
You suspect the above diagnoses are related to his current condition and you move on to undertake some further investigations
What tests might be helpful to confirm the diagnosis?
Whilst myoglobin is raised in rhabdomyolysis in someone like this gentleman, where the disease is already progressed, it is likely that this will no longer show in the urine due to blockage in the renal tubule.
This would be unusual to find in a patient with rhabdomyolysis. If there is significant haemolysis, this would lead to an increase in unconjugated bilirubin hover the most common liver enzyme derangement in rhabdomyolysis is ALT.
This tends to be markedly elevated and is the most specific test in rhabdomyolysis and we would expect this to be raised in this gentleman’s case.
Whilst myoglobin is raised in rhabdomyolysis in someone like this gentleman, where the disease is already progressed, it is likely that this will no longer show in the urine due to blockage in the renal tubule.
Blood pressure can be abnormal for a whole variety of reasons and as such is not a specific indicator of Rhabdomyolysis.
You carry out the investigations that we have agreed on above
ECG – Tented T- waves and broad QRS complexes
CK : 3689U/L
Urine myoglobin – positive
Given the results that you have for your patient you decide to instigate treatment for this individual.
What will be important in this gentleman’s management?
Select all options that apply
In most cases of rhabdomyolysis IV fluids will form a part of the management of this condition. This needs to be used with caution in those individuals with significant renal impairment who are anuric or oligouric and thus there is a risk of fluid overload leading to peripheral or pulmonary oedema
While IV bicarbonate has previously been used in the management of rhabdomyolysis (little evidence that this is of benefit) oral bicarbonate is certainly not involved in the management of rhabdomyolysis.
In some individuals, impairment to their kidneys will be so severe that dialysis will be required. Remember indications for emergency dialysis – hyperkalaemia which is not responsive to medical management, pulmonary oedema, progressive uraemic encephalopathy.
Whilst not a specific treatment for Rhabdomyolysis it is important to treat hyperkalaemia in this gentleman. As he has cardiac changes related to hyperkalaemia, calcium gluconate is important to stabilise the cardiac membrane and prevent progression to deadly arrhythmias.
This gentleman is mildly anaemic however not to an extent to where he would require blood products. A potential complication of rhabdomyolysis is DIC and should this become an issue then blood products may be required, however there is no indication that this would be part of the management at this time.
You decide a management plan and discuss it with your senior who agrees.
This gentleman is managed in a ICU setting and with the measures you have decided on he and his renal function gradually improves over the next 9 days.
When he was discharged, his renal function was in a normal range for his age.
Well done you have completed this case.
