Diabetic nephropathy is the kidney disease that occurs as a result of diabetes. It is a leading cause of kidney failure in Europe and the USA. After many years of diabetes the delicate filtering system in the kidney becomes destroyed, initially becoming leaky to large blood proteins such as albumin which are then lost in urine. This is more likely to occur if the blood sugar is poorly controlled.
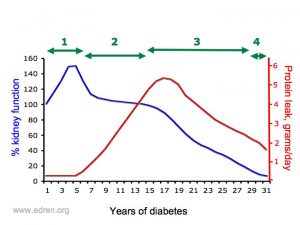 |
| The diagram shows how kidney function reduces and the amount of protein in the urine increases in diabetic nephropathy. |
Diabetic kidney disease typically follows this pattern over many years. Treatment can slow and sometimes stop it from moving on to the next stage, especially early on:
1. It begins with a tiny amount of protein appearing in the urine – this is called microalbuminuria. The kidney function is sually normal at this point.
2. Over 10-15 years protein in urine (proteinuria) increases, and nephrotic syndrome may develop
3. As the levels of proteinuria rise, the kidneys begin to work less well. The eGFR, which is very approximately equivalent to % kidney function, falls over years.
4. Eventually it reaches “end-stage renal disease” (ESRD), when dialysis or a kidney transplant is needed to keep you alive.
Do all diabetics develop nephropathy?
The overall risk of developing diabetic nephropathy varies between about 10% of type II diabetics (diabetes of late onset) to about 30% of type I diabetics (diabetes of early onset). There are many factors, some known and others not, that affect the individual risk of developing diabetic nephropathy. These things are some of those known to increase the risk of getting diabetic nephropathy:
- Poor blood sugar control
- High blood pressure
- Smoking
- Relatives have had kidney disease or high blood pressure
- Diabetes began in teens
- Male
- Indo-Asian or Afro-Carribean background
Will I need a kidney biopsy?
If your kidney disease has followed the typical pathway shown above, and there are other signs of diabetic complications, a kidney biopsy is usually not needed. If there are unusual features, then further investigations may be needed to be sure – this sometimes includes a kidney biopsy.
 What else happens in diabetic nephropathy?
What else happens in diabetic nephropathy?
High blood pressure is often the first sign of diabetic nephropathy.
Other ‘microvascular’ complications – diabetic nephropathy is one of the ‘microvascular’ complications of diabetes, meaning disease of tiny arteries. It is common for patients with nephropathy to also have signs of diabetic eye disease, and some will also have diabetic neuropathy.
Large blood vessel disease – people with microvascular complications of diabetes are also prone to get accelerated atherosclerosis, narrowing of larger arteries. Heart attacks, strokes, and narrowing of blood vessels to the legs occur more often and at a younger age than usual.
What can be done to reduce the risk of problems?
Blood glucose control: Good blood glucose control can prevent the development and slow the progression of diabetic nephropathy, as well as preventing the other complications of diabetes. This can not be achieved by tablets and/or insulin alone, but requires a good diet too.
Blood pressure control: The recommended target blood pressure is 125/75 mmHg in diabetic patients however, the lower the blood pressure the lower the risk of problems. This usually requires more than one type of tablet to achieve. If you are overweight, losing weight will help too.
ACE inhibitors and Angiotensin Receptor Blockers (ARBs): Angiotensin-Converting Enzyme (ACE) inhibitors and Angiotensin Receptor Blockers (ARBs) lower blood pressure, but also have an additional protective effect on diabetic kidneys.
Newer drugs to prevent diabetic nephropathy: several newer anti-diabetic drugs seem to have additional protective effects. The best established at present are the SGLT2 inhibitors.
Diet: Above and beyond the diabetic diet, not adding any salt, and reducing alcohol intake will have beneficial effects on blood pressure. Other aspects of diet (including energy, calcium and phosphate, and protein) can be important in advanced CKD is severe (e.g. stage 4+).
Controlling blood fat and cholesterol: Controlling blood fat and cholesterol levels helps prevent heart disease and possibly strokes, and may slow the progression of diabetic kidney disease. The current data points towards a target total cholesterol of <3.5mmol/l if you have microalbuminuria.
Smoking: You really shouldn’t smoke, not only for the sake of your kidneys, but also for the sake of your heart and brain blood vessels. Smokers die earlier than non-smokers, but diabetic smokers die much earlier and often develop serious circulation problems at a young age.
What is the treatment?
|
Stage |
Assessment | Treatment |
| No proteinuria | Monitor blood pressure (BP) Monitor blood glucose Screen for microalbuminuria if type I diabetic for over 5yrs, or type II diabetic |
Aim under 130/80mmHg (120/70mmHg in type I diabetes). Aim HbA1c under 7% Dietary advice for sugar and fat STOP SMOKING |
| Microalbuminuria | Close monitoring of blood pressure, blood glucose, and blood lipids. Monitor urinary protein |
Aim BP <125/75mmHg Add further blood pressure lowering drugs if needed. Add ACEi/ARBif possible. Aim total cholesterol under 3.5mmol/l Add ACE inhibitor if possible |
| Proteinuria | Close monitoring of blood pressure, blood glucose, and blood lipids. Monitor urinary protein |
As for microalbuminuria |
| Declining kidney function | Prepare for dialysis and/or transplantation |
Further information
- High blood pressure and kidney disease (edren, this site).
- Chronic renal failure and its progression (edren, this site).
- National Institute of Diabetes and Digestive and Kidney disease (USA) has good information on diabetes, including nephropathy (kidney disease), retinopathy, neuropathy.
- Diabetes UK provides information and patient support about diabetes more generally.
Key Points
- Diabetic nephropathy does not occur in all diabetics.
- The risk of diabetic nephropathy is greater when the control of blood glucose is poor, as well as in those diabetic patients who have high blood pressure and are smoker.
- Diabetic nephropathy is a sign of worsening blood vessel disease throughout the body, and is associated with an increased risk of heart attack, stroke and circulatory problems.
- The risk and effect of diabetic nephropathy can be reduced by controlling blood sugar, blood pressure and cholestrol levels. This can be achieved with a combination of good dietary control as well as medication.
- Smoking shortens life. You should stop!
Acknowledgements: The original author of this page was Walaa Saweirs. It was substantially reviewed in May 2020. The date it was last modified is shown in the footer.
