Contents
Overview
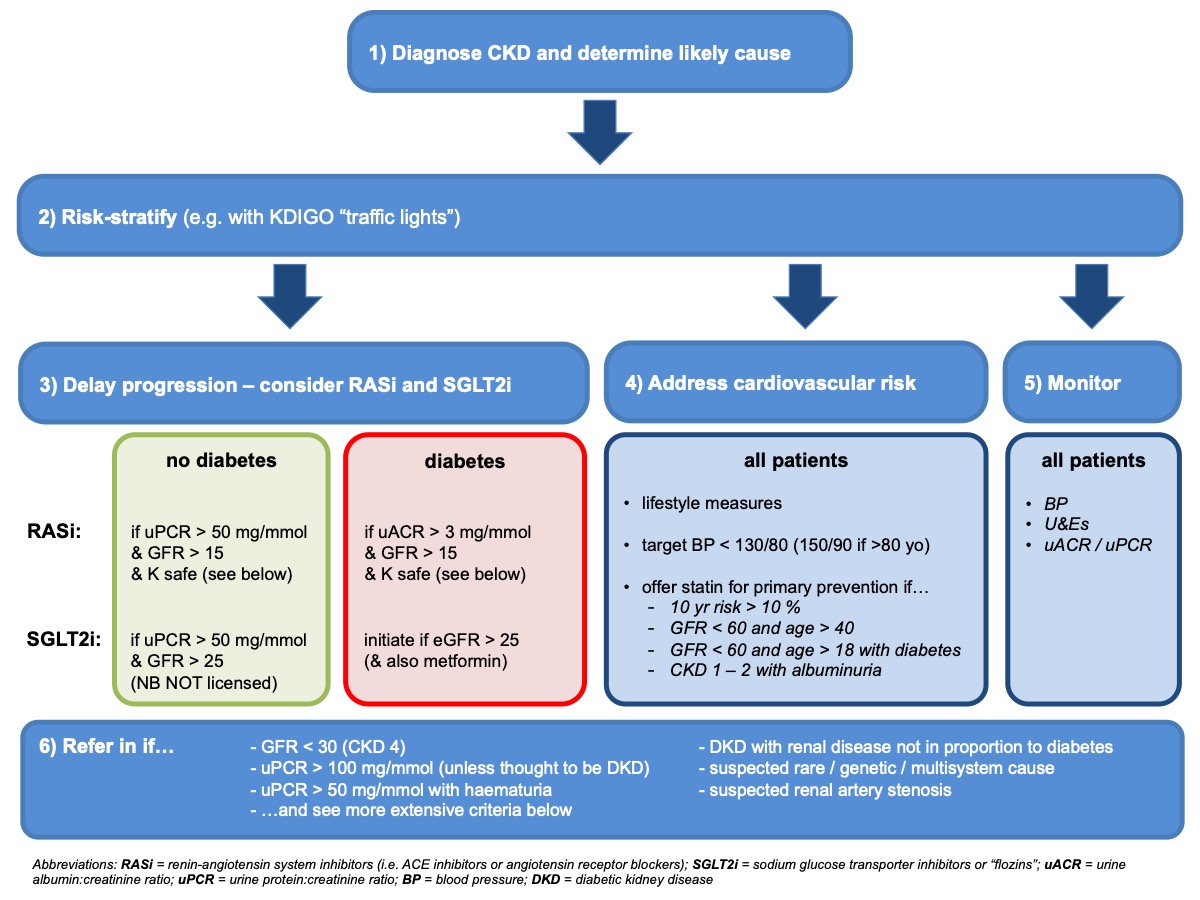
Most chronic kidney disease (CKD) is diagnosed and managed in primary care. The advice on this page is based largely on consensus CKD guidelines from KDIGO and NICE and diabetes guidelines from KDIGO. A general approach is summarised here. Further details on each of the sections of this diagram are given below:
1) Diagnose CKD and determine likely cause
Definition of CKD
CKD is defined by KDIGO as “abnormalities of kidney structure or function, present for at least 3 months, with implications for health”. Therefore the new finding of an elevated serum creatinine may be CKD or an acute kidney injury.
CKD is staged according to eGFR and albuminuria. Some patients with structural abnormalities (e.g. cysts) may have CKD despite normal eGFR and no albuminuria.
Proteinuria
Checking for proteinuria is important in:
- diagnosing CKD (some patients can have proteinuria as the only sign of CKD – i.e. with normal GFR and kidney imaging; for example this is the case in many patients with nephrotic syndrome; it is generally unhelpful to screen for proteinuria in an unselected, asymptomatic population)
- risk-stratification to guide management (e.g. patients with proteinuria may benefit from ACE/ARB to delay CKD progression)
- risk-stratification to guide prognostication (e.g. patients with proteinuria are at greater risk of cardiovascular disease and CKD progression)
- monitoring CKD progression / response to treatment
For further information on how to check for proteinuria and how to interpret results, see here.
Determining the cause of CKD
The commonest causes of CKD in Scotland are vascular disease and diabetes. There are of course many other causes however, so it is worth considering the likely aetiology and in particular ensuring that reversible causes are not missed. Look for features of multi-system disease and review all medications, including over-the-counter medications such as NSAIDs and herbal remedies.
Important diagnoses not to miss include:
- urinary tract obstruction
- drug causes of interstitial nephritis / scarring (NSAIDs, 5-asa drugs, rarely PPIs, lithium)
- paraprotein-related kidney disease
- multisystem inflammatory disease (vasculitis, lupus)
Consider a renal USS if (NICE guidelines):
- accelerated progression of CKD
- visible or persistent invisible haematuria
- symptoms of urinary tract obstruction
- family history of polycystic kidney disease and are aged over 20 years
- GFR < 30 ml/min/1.73 m2 (GFR category G4 or G5)
- …and consider the likely utility of scanning in context of age / frailty / co-morbidities etc. (not everybody with CKD needs an USS!)
2) Risk-stratify
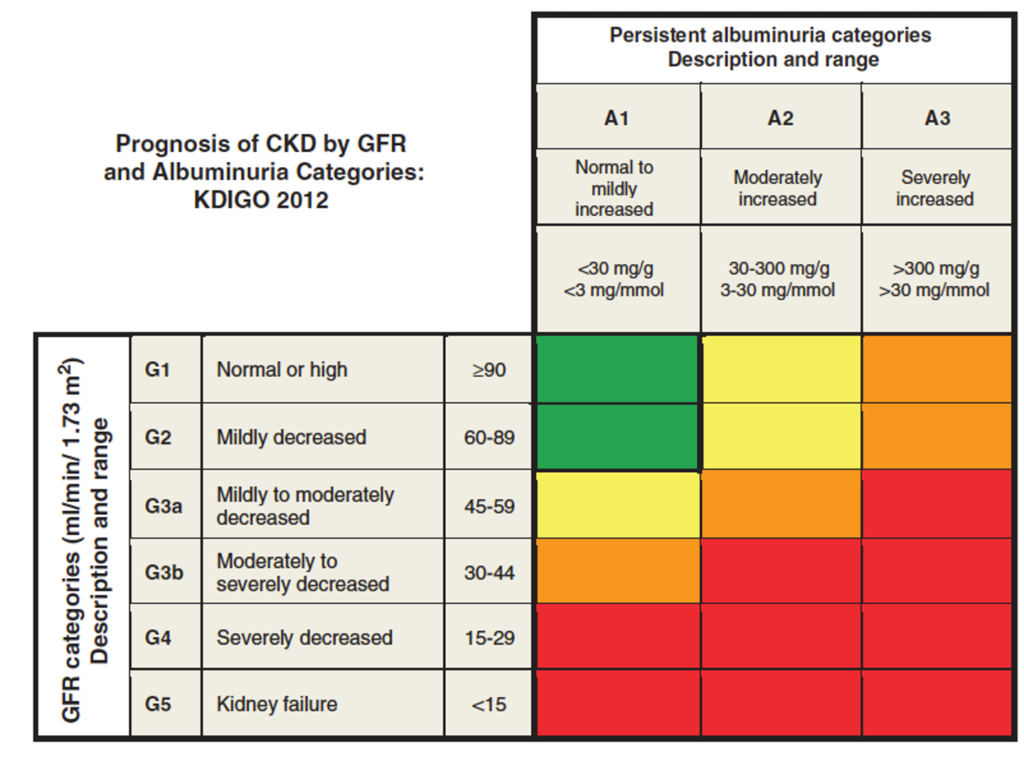
Management is dictated by risk of CKD progression. This can be assessed using the KDIGO “traffic light” grading of GFR and albuminuria:
Patients in the orange and red categories are at high risk of progression.
Other methods of risk stratification include:
- looking at historic trends in serum Cr / eGFR (ideally on a graph if possible)
- using the kidney failure 4-variable risk equation
- factoring in what is known about the natural history of the underlying kidney disease (e.g. diabetic kidney disease is often progressive)
3) Delay CKD progression
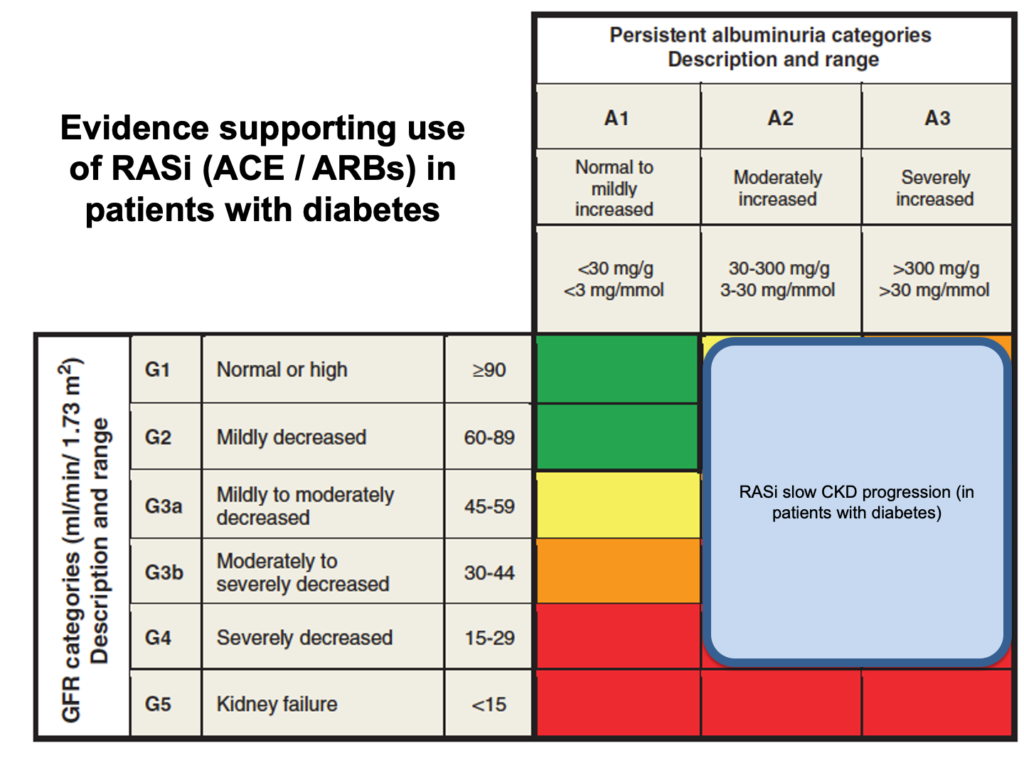
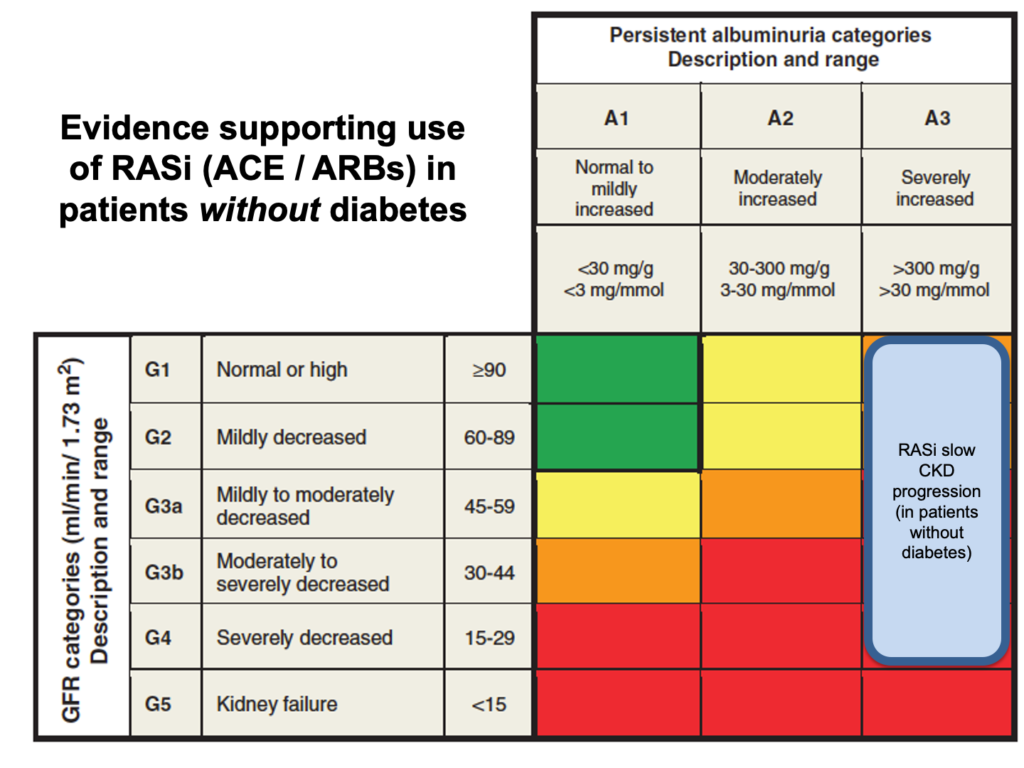
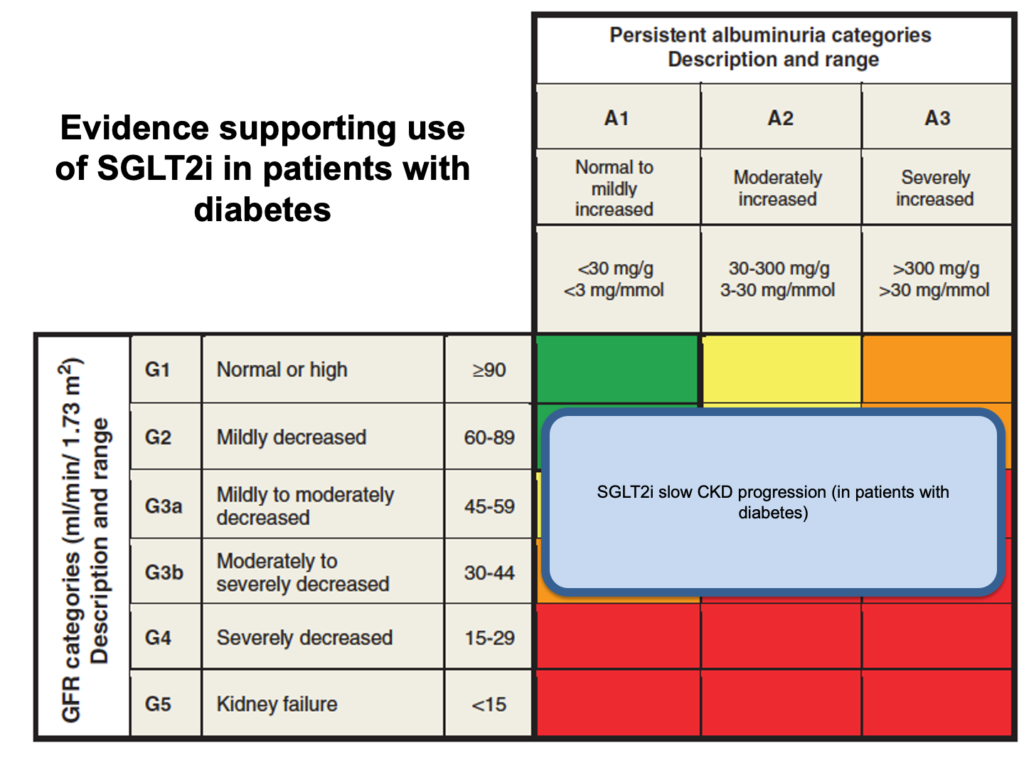
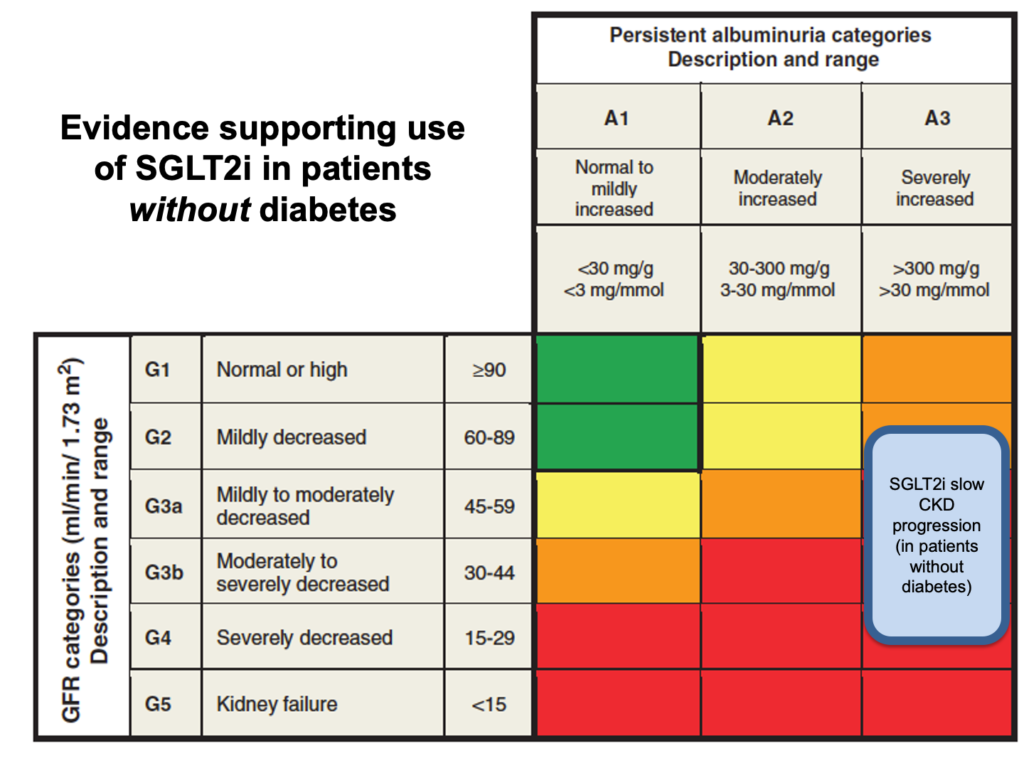
In some CKD subgroups, renin-angiotensin system inhibitors (RASi – i.e. ACE inhibitors or angiotensin receptor blockers) and SGLT2i have been shown to slow progression of CKD. The patients in whom there is an evidence base to support these drugs is shown here:
RASi have been shown to reduce the risk of end-stage renal failure by ~30% (i.e. relative risk reduction 0.3). In patients already established on RASi, SGLT2i have been shown to reduce the risk of renal progression and death by ~40% (i.e. relative risk reduction 0.4) – a benefit that is realised even in patients without diabetes. The absolute risk reduction (ARR) depends on the starting risk. Therefore patients with low risk of progression to ESKD (e.g. green on the KDIGO risk table) will have a low ARR and a NNT that is measured in the high-100s to 1000s. Patients with a moderate-to-high baseline risk of progression (KDIGO orange / red) stand to benefit enormously from these drugs and would have NNTs in the 10s to low-100s.
We recommend considering these drugs in patients at risk of progression, taking into account other factors such as:
- patient preference
- other co-morbidities (e.g. in heart failure, RASi and SGLT2i might confer an ever greater mortality benefit)
- effect on cardiovascular risk (i.e. not just effect on CKD progression)
- glycaemic control in diabetes
- polypharmacy
- competing risk of death from other causes (e.g. elderly patients with CKD are more likely to die from non-renal causes before reaching end-stage renal failure; of course they may still benefit from the cardiovascular effects of RASi)
In draft guidance:
- NICE recommend dapagliflozin 10 mg od for CKD as an add-on to ACEi/ARB (or if ACEi/ARB not tolerated) and if eGFR 25 – 75 ml/min and uACR > 22.6 mg/mmol (in NHSL we have taken 25 mg/mmol as a convenient, user-friendly threshold)
- the UKKA recommend SGLT2i in patients with CKD and uACR > 25 mg/mmol, excluding individuals with ADPKD or on immunosuppressive medication.
RASi and SGLT2i algorithms
For patients without diabetes:
- consider RASi if uACR > 30 mg/mmol (uPCR > 50) and hypertension
- consider RASi if uACR > 70 mg/mmol (uPCR > 100) regardless of blood pressure
- consider SGLT2i if uACR > 25 mg/mmol (uPCR > 50) in patients who are already established on maximum-dose RASi (or if RASi contraindicated)
For patients with T2DM not on insulin:
- consider RASi if uACR > 3 mg/mmol regardless of blood pressure
- consider SGLT2i in all patients with CKD (i.e. if eGFR < 60 or uACR > 3)
- consider metformin in all patients with CKD and eGFR > 30 ml/min (as per BNF)
For patients with T1DM or T2DM on insulin:
- consider RASi if uACR > 3 mg/mmol regardless of blood pressure
- discuss use of SGLT2i with diabetes team (risk of euglycaemic ketoacidosis)
Prescribing RASi
It is unusual to start RASi if serum potassium > 5 mmol/l or eGFR < 15. However, for patients who stand to gain a lot (e.g. diabetic kidney disease with albuminuria) it is often appropriate to tolerate a higher serum potassium (e.g. up to 6 mmol/l) and to consider strategies to lower potassium (dietary restriction, diuretics, novel binders). Discuss with renal if unsure. We have more advice on our ACE inhibitor page.
Prescribing SGLT2i
See our page on initiating SGLT2i in patients with and without diabetes. SGLT2i for CKD are now approved by the SMC and on the NHS Lothian formulary.
4) Address cardiovascular risk
Patients with CKD have elevated cardiovascular risk. In addition to standard measures that apply to the general population, consider offering atorvastatin (starting dose 20 mg) if:
- GFR < 60 and age > 40
- GFR < 60 and age > 18 with diabetes
- CKD 1 – 2 with albuminuria
Increase to 40 mg daily if not achieving appropriate lipid targets (e.g. NICE recommend if reduction in non-HDL cholesterol < 40%) and eGFR > 30.
Guidelines differ in their recommended target blood pressures. In most patients (with or without CKD) it is usually appropriate to target an office blood pressure of 130/80 or 150/90 in the frail elderly. See our blood pressure pages for a more nuanced approach.
5) Monitor
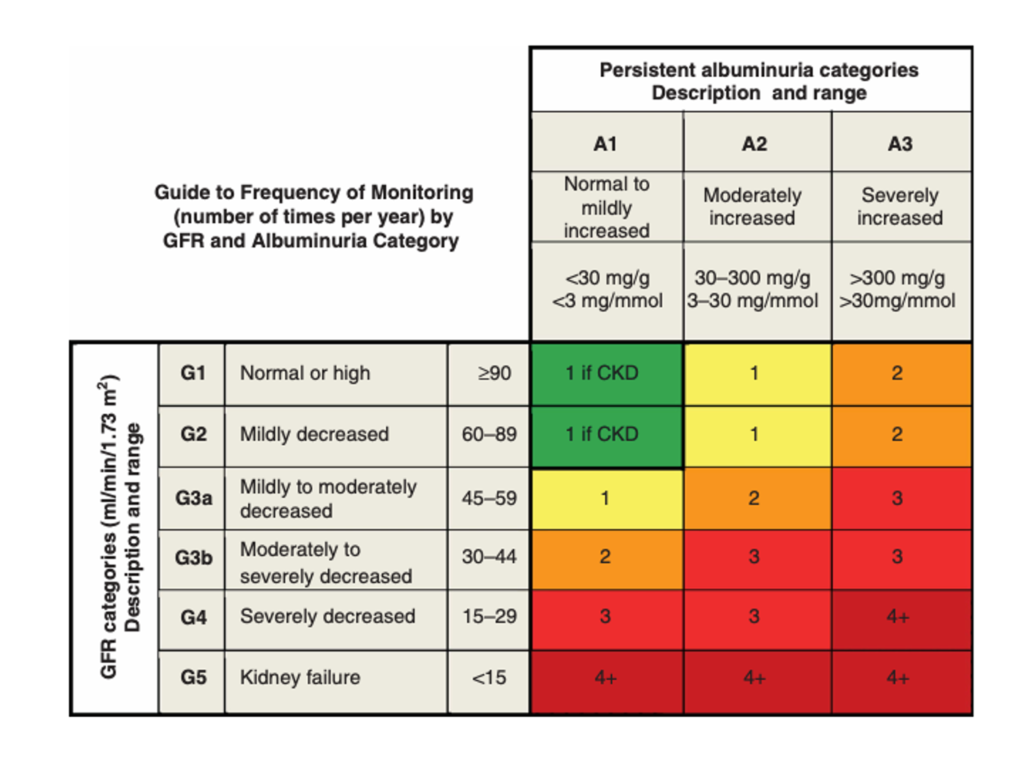
Patients with CKD should have blood pressure, U&Es and proteinuria (e.g. uACR) monitored. This figure suggests monitoring frequency (with the numbers representing the number of tests per year):
It is sensible to adjust this according to patient preferences / characteristics. For example, patients opting for predominantly conservative management may not need any monitoring at all. Newly diagnosed patients may need more frequent monitoring, until stability has been established.
6) Referral to renal
See our referral guidelines. Some common indications for referral in CKD are given in the summary figure at the top of this page. If you have questions regarding the diagnosis or management of CKD that may not warrant formal referral to the renal clinic, please make use of our e-mail advice line (rie.renaladvice@luht.scot.nhs.uk). Please note that this advice line is for health professionals seeking advice about patients in Lothian & Borders.